 Previous experimental data from our laboratory suggest that PAMAM dendrimers are a useful scaffold that can be coupled to make polymeric SA conjugates that will inhibit influenza hemagglutinin protein binding at reduced concentrations. They posses multiple-surface functional groups that can provide an attachment site for SA. 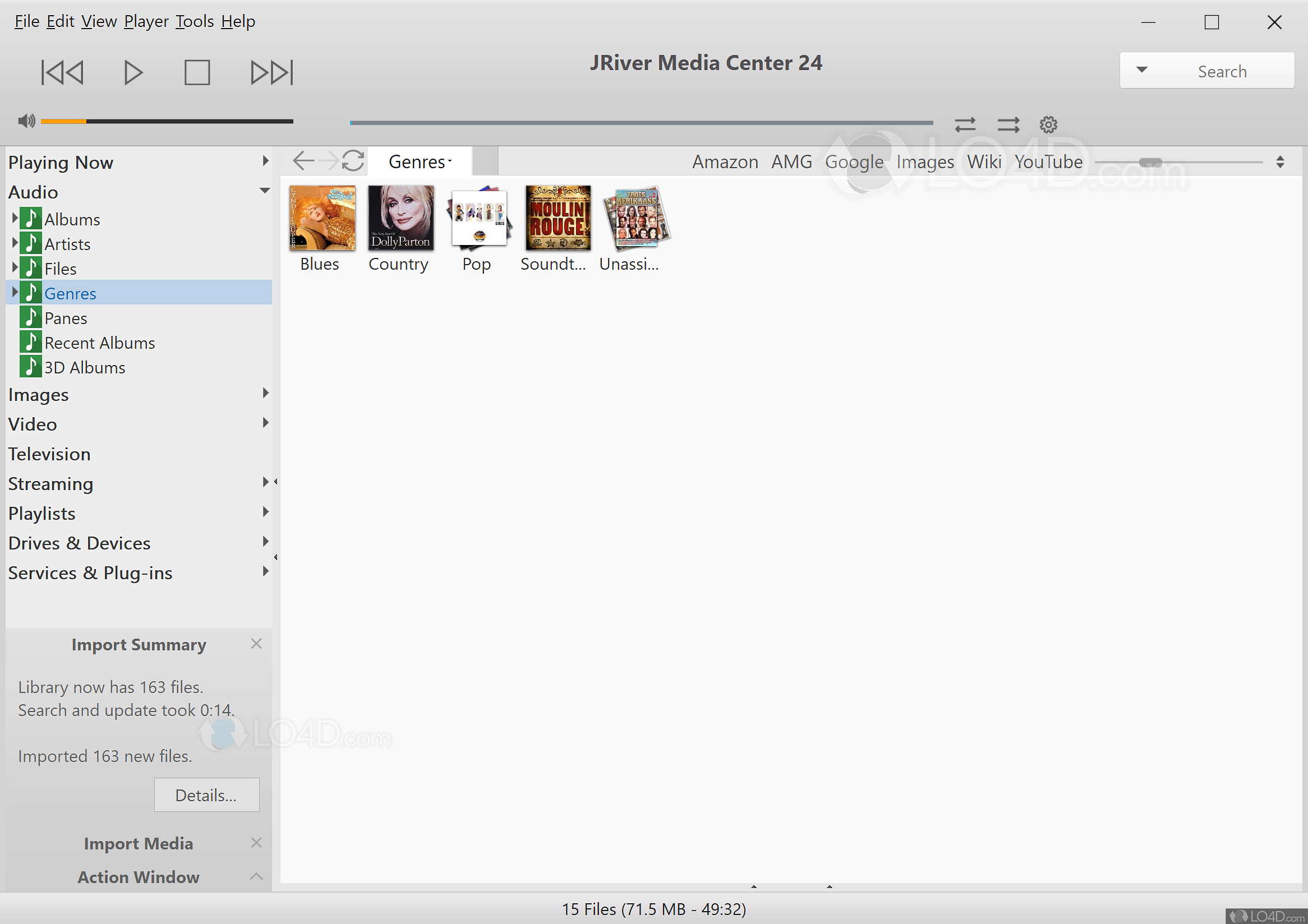
Polyamidoamine (PAMAM) dendrimers are monodispersed, water soluble, macromolecules that are highly branched and well defined. A potential solution could involve conjugating SA molecules to a macromolecule to increase delivery of SA while reducing cytotoxicity. Thus, high concentrations of monomeric SA must be delivered to inhibit viral infection, and these concentrations are toxic. However, these monomers become susceptible to rapid enzymatic breakdown in vivo and do not effectively compete with polymeric sialosides on host cells. 
C Generation 4 (G4) SA-conjugated polyamidoamine (PAMAM) dendrimerĪ decoy approach to prevent influenza infection might use monomeric SAs or methyl sialosides to block viral adhesion to cells. This binding action is the crucial component for the initiation of infection and, therefore, serves as a potential target to decoy therapy with effectiveness across different subtypesĪ Sialic acid (SA) unit. Sialic acid (SA) molecules present on cellular surface structures (glycoproteins or glycolipids) are the targets for binding by hemagglutinin ( figure 1). 
Antigenic drift mutations and antigenic shift of the gene segments that encode hemagglutinin and neuraminidase contribute to the preservation of this virus and present problems for vaccine and small-molecule therapeutic development. Hemagglutinin consists of 2 structural domains, with the peripheral, globular domain having a binding cleft for attachment to cells. Segment 4 of the influenza A genome encodes the major surface glycoprotein, which is called hemagglutinin because of its ability to agglutinate erythrocytes. These viruses have evolved some exceptionally effective survival mechanisms and are able to circumvent the immune response because of the highly mutable genes that encode their surface proteins. Annual epidemics and occasional pandemics of influenza A virus are a significant health concern. Influenza A virus is an enveloped virus with a segmented, single-stranded, RNA genome. This binding may be inhibited by an extracellular therapeutic agent that resembles the surface-binding component of the host. Theoretically, blocking the initial interaction of a virus with this host cell receptor can prevent viral infection. Most viruses use a receptor that binds to a cellular surface component as a targeting mechanism. 
Polyvalent binding inhibitors have potential as antiviral therapeutics, but issues related to strain specificity must be resolved In vivo experiments showed that G4-SA completely prevented infection by a H3N2 subtype in a murine influenza pneumonitis model but was not effective in preventing pneumonitis caused by an H2N2 subtype. In contrast, G4-SA had no ability to inhibit hemagglutination with H2N2 subtypes or 2 of 5 H1N1 subtype strains. In hemagglutination-inhibition assays, G4-SA was found to inhibit all H3N2 and 3 of 5 H1N1 influenza subtype strains at concentrations 32–170 times lower than those of SA monomers. Polyvalent, generation 4 (G4) SA-conjugated polyamidoamine (PAMAM) dendrimer (G4-SA) was evaluated as a means of preventing adhesion of 3 influenza A subtypes (H1N1, H2N2, and H3N2). Free SA monomers cannot block hemagglutinin adhesion in vivo because of toxicity. Influenza A viral infection begins by hemagglutinin glycoproteins on the viral envelope binding to cell membrane sialic acid (SA). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
